On Thursday, May 14, 2026, the NIDA IRP Clinical Program welcomed representatives from the Office of the Lieutenant Governor. Aruna Miller and her staff received a tour of our facilities and learned about the cutting edge research we are conducting in the field of addiction medicine.
News Main
Technology Resource Initiative – Paper of the Month – May 2026
Cocaine chemogenetics blunts drug-seeking by synthetic physiology. Nature
Cocaine-gated chemogenetics enables selective modulation of addiction-related circuits while sparing natural reward.
Chronic delivery of buprenorphine during abstinence decreases incubation of heroin seeking and neuronal activation in medial prefrontal cortex and striatum in male and female rats
Featured Paper of the Month – May 2026
Published in JAMA Network Open by Xiang Xiao and Yihong Yang et al. of the NIDA IRP Magnetic Resonance Imaging and Spectroscopy Section.
Buprenorphine is an FDA-approved medication for opioid addiction, but the brain regions underlying its therapeutic effects are unknown. In this study, we tested whether chronic buprenorphine reduces the time-dependent increase (“incubation”) in heroin seeking during abstinence.
Persistent representation of a prior schema in the orbitofrontal cortex facilitates learning of a conflicting schema
Hot Off the Press – April 16, 2026 Published in Nature Communications by Ido Maor and Geoffrey Schoenbaum, et al. of the NIDA IRP Behavioral Neurophysiology Neuroscience Section. Summary When Old Knowledge Helps Learning Something New, even When It Conflicts How does the brain use past experience to guide learning in new situations, especially when… [Read More]
NIH IRP GLP-1 Workshop
Friday, July 24, 2026 – 8:30 am – 4:15 pm Masur Auditorium – Building 10 NIH Bethesda Campus NIH Videocast Link Here Chairs/Organizers Lorenzo Leggio, M.D., Ph.D., NIDA/NIAAA Mike Krashes, Ph.D., NIDDK Schedule 8:45 – 8:55 Lorenzo Leggio, M.D., Ph.D. (NIDA/NIAAA) and Mike Krashes, Ph.D. (NIDDK) Welcome 8:55 – 9:00 Janice Lee, D.D.S., M.D., M.S., Deputy… [Read More]
Modification of Brain Connectome on Association Between Adverse Childhood Experiences and Development of Mental Disorders in Preadolescence
Featured Paper of the Month – April 2026
Published in JAMA Network Open by Xiang Xiao and Yihong Yang et al. of the NIDA IRP Magnetic Resonance Imaging and Spectroscopy Section.
In this study, we addressed the question of whether a brain marker moderates the association between childhood adversity and psychiatric disorders during the transition to adolescence.
Predicting individual incubation of opioid craving by whole-brain functional connectivity
Hot Off the Press – March 18, 2026 Published in PNAS by Ying Duan, Pei-Jung Tsai and Yihong Yang, et al. of the NIDA IRP Magnetic Resonance Imaging and Spectroscopy Section. Summary This study is a trans-branch collaboration between groups of Drs. Yihong Yang and Yavin Shaham at NIDA-IRP. Relapse driven by craving remains a challenge in… [Read More]
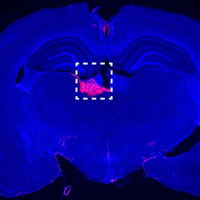
Cocaine chemogenetics blunts drug-seeking by synthetic physiology
Hot Off the Press – March 9, 2026 Published in Nature by Juan Gomez and Michael Michaelides et al. of the NIDA IRP Biobehavioral Imaging and Molecular Neuropsychopharmacology Section. Summary Our research team is pleased to announce the publication of our latest study in Nature, introducing a transformative synthetic biology platform designed to study and… [Read More]
Technology Resource Initiative – Paper of the Month – March 2026
Long-term labeling and imaging of synaptically connected neuronal networks in vivo using double-deletion-mutant rabies viruses Nat Neurosci
Rabies-virus-based monosynaptic tracing is a common tool used in neuroscience research; however, toxicity of the virus limits the duration of experimental observations. Jin et al. have produced a second generation system for long-term monosynaptic tracing using a double-deletion-mutant rabies virus that reduces this toxicity and allows for viral replication and spread through neuronal subsets.
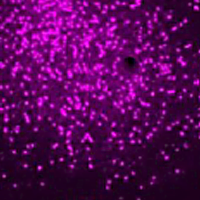
Distinct Amygdala Neuronal Populations Control Opioid Use and Withdrawal in Mice.
Featured Paper of the Month – March 2026
Published in Biological Psychiatry by Lucas Silva Tortorelli and Leandro Vendruscolo et al. of the NIDA IRP Stress and Addiction Neuroscience Unit.
We used in situ hybridization to characterize the expression of μ opioid receptor (MOR) (Oprm1), and we used behavioral and molecular approaches to assess the functional role of these CeA neuronal populations in opioid-dependent mice.