Hot Off the Press – June 7, 2021
Published in eLife by Justin Siemian and Yeka Aponte, et al. in the NIDA IRP Neuronal Circuits and Behavior Unit.
Decades of work have revealed the lateral hypothalamus (LH) as a crucial region for orchestrating appetitive and reward-related behaviors. However, the contributions of genetically-identified lateral hypothalamic neurons to other survival processes such as nociception have not been unraveled.
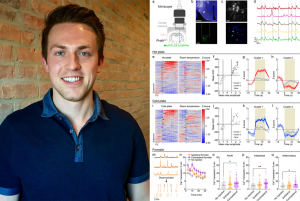
In this manuscript, Siemian et al. use a combination of approaches to identify, selectively target, monitor, and manipulate the activity of a small population of glutamatergic parvalbumin-positive lateral hypothalamic (LHPV) neurons and their projections to study their involvement in a broad range of pain modalities. Using in vivo functional imaging, they show for the first time that LHPV neurons exhibit an array of time-locked responses to noxious events. Moreover, chemogenetic activation of LHPV neurons not only reduces traditional pain-stimulated behaviors but also restores pain-suppressed behavior and reduces pain-associated negative affect. In models of persistent inflammatory or neuropathic pain, optogenetic activation of LHPV neurons or their axonal projections in the ventrolateral periaqueductal gray area (vlPAG) attenuates nociception, and rabies virus-mediated neuroanatomical tracing reveals that LHPV neurons preferentially target glutamatergic neurons over GABAergic neurons in the vlPAG. By contrast, LHPV axonal projections to the lateral habenula (LHb) regulate aversion but not nociception. Finally, they find that LHPV neuronal activation evokes additive to synergistic antinociceptive interactions with morphine and restores morphine antinociception following the development of morphine tolerance. Their findings identify LHPV neurons as a lateral hypothalamic cell type intricately involved in nociception and demonstrate their potential as a novel target for pain treatment or for use in combination therapies with current analgesics.
Publication Information
An excitatory lateral hypothalamic circuit orchestrating pain behaviors in mice Journal Article
In: eLife, vol. 10, pp. e66446, 2021, ISSN: 2050-084X.